 
Two other hydrates are known, pale green Cl 2 It consists of the cation trans- + and additional molecules of water and a chloride anion in the lattice. The common hexahydrate can be more precisely described as Cl♲H 2O. Several members are known of the series of q+. The different forms exist both as solids and in aqueous solutions. 
The hydrated chromium(III) chlorides display the somewhat unusual property of existing in a number of distinct chemical forms (isomers), which differ in terms of the number of chloride anions that are coordinated to Cr(III) and the water of crystallization. If pressurized to 9.9 GPa it goes under a phase transition. The anhydrous CrCl 3 is exfoliable down to the monolayer limit. For this reason, crystals of CrCl 3 cleave easily along the planes between layers, which results in the flaky ( micaceous) appearance of samples of chromium(III) chloride. 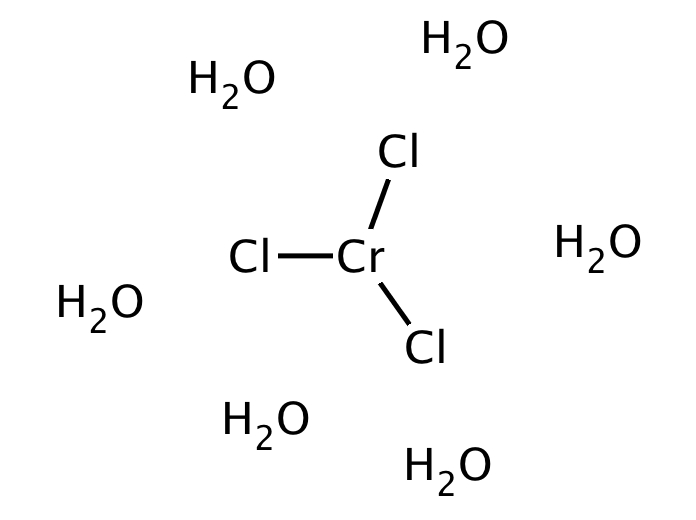
The absence of cations in alternate layers leads to weak bonding between adjacent layers. Chromium chlorides find use as catalysts and as precursors to dyes for wool.Īnhydrous chromium(III) chloride adopts the YCl 3 structure, with Cr 3+ occupying one third of the octahedral interstices in alternating layers of a pseudo- cubic close packed lattice of Cl − ions. The anhydrous compound with the formula CrCl 3 are violet crystals, while the most common form of the chromium(III) chloride are the dark green crystals of hexahydrate, CrCl 3♶H 2O.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
